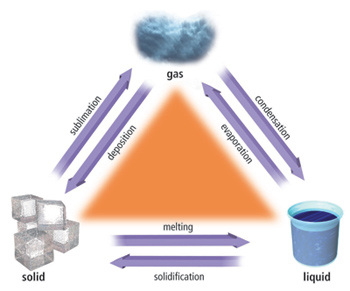
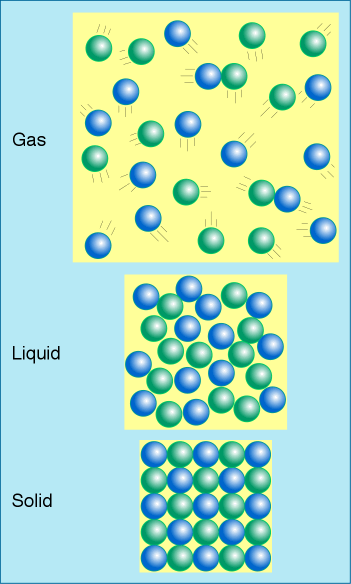
You should already know what melting and freezing are. However, you also need to know what sublimation, deposition, evaporation, and condensation are as well.
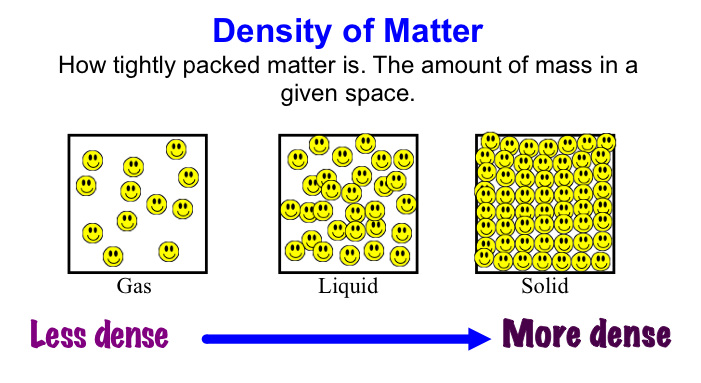
DENSITY
What is density? It is mass over volume. Solid should be more dense than liquid and gas. Why? Because the particles are packed together, their volume is smaller, making the density higher.
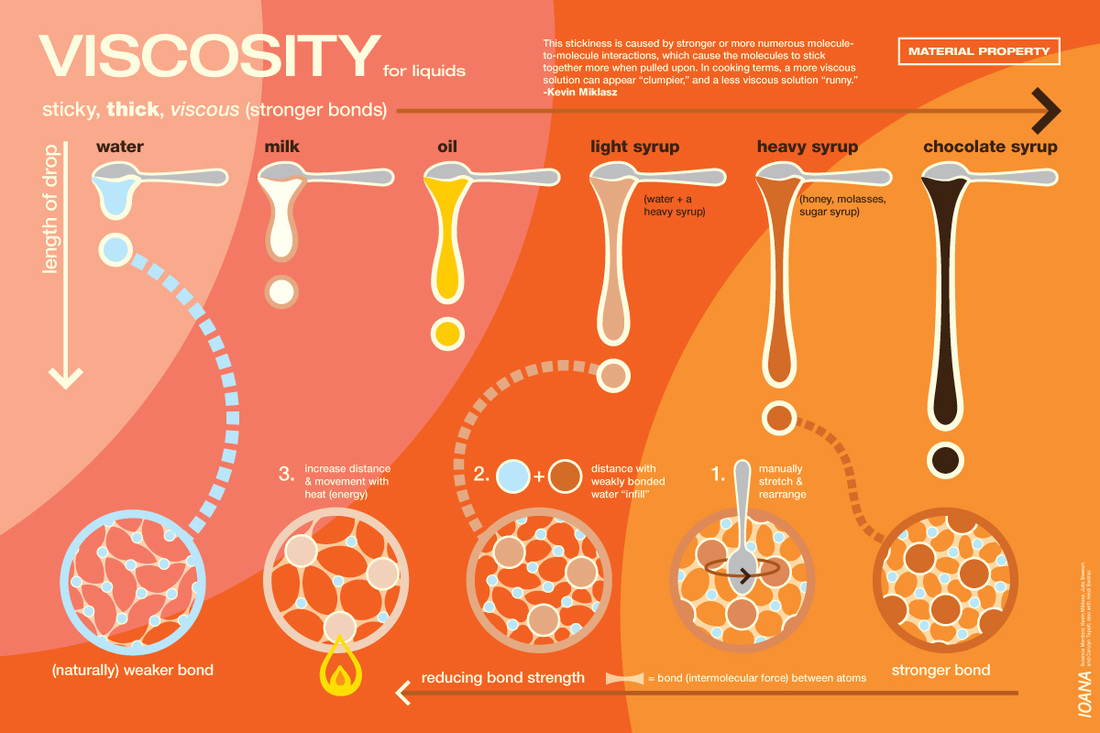
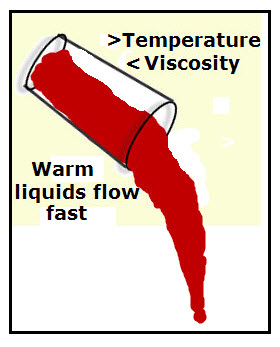
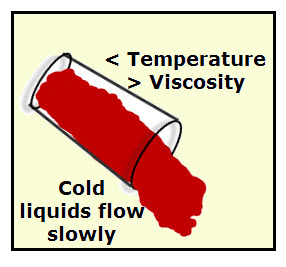
VISCOSITY
Viscosity is the resistance to flow. The higher the viscosity, the higher the resistance to flow. That means it's going to be slower to flow.
Viscosity is the resistance to flow. The higher the viscosity, the higher the resistance to flow. That means it's going to be slower to flow.
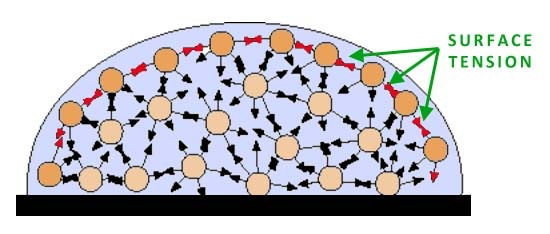
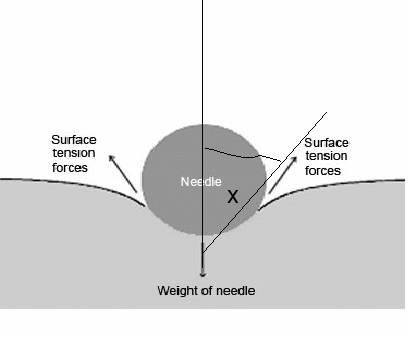
SURFACE TENSION
Surface tension is the attractive force between particles in a liquid that minimizes surface area. This is due to hydrogen bonding which is a strong force. Since all the forces are in the middle, it makes a spherical shape.
Surface tension is the attractive force between particles in a liquid that minimizes surface area. This is due to hydrogen bonding which is a strong force. Since all the forces are in the middle, it makes a spherical shape.
Cohesion is the force of attraction between identical molecules in a liquid (cohesion is a result of intermolecular forces).
Adhesion is the force of attraction between liquid molecules and a solid that is touching them.
Meniscus of water in a glass tube is concave: adhesion > cohesion
Meniscus of Hg in a glass tube is convex: cohesion > adhesion
Crystalline - repeating geometric pattern
Amorphous – no geometric pattern
Adhesion is the force of attraction between liquid molecules and a solid that is touching them.
Meniscus of water in a glass tube is concave: adhesion > cohesion
Meniscus of Hg in a glass tube is convex: cohesion > adhesion
Crystalline - repeating geometric pattern
Amorphous – no geometric pattern