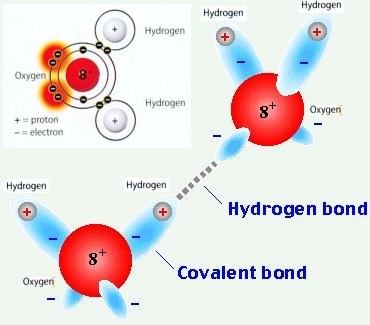
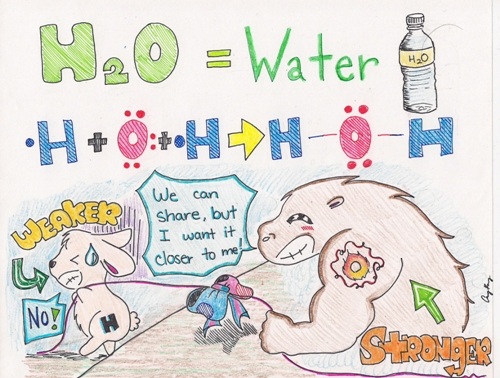What is a covalent bond?
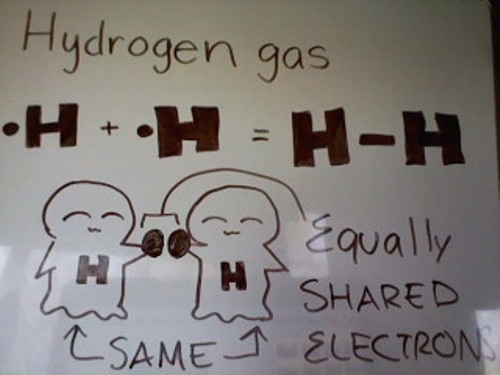
It is the sharing of electrons!
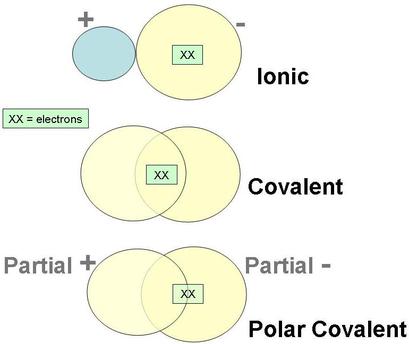
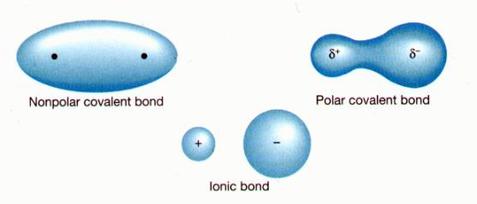
Sometimes the electrons are shared equally. When they are shared equally, we use the term nonpolar. Nonpolar - electrons are shared EQUALLY.
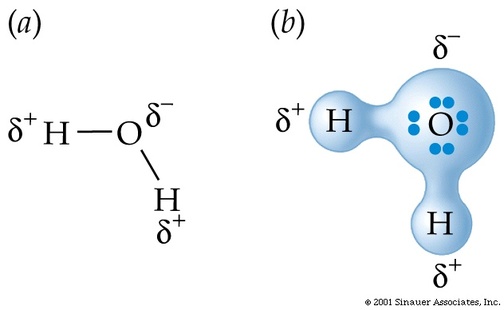
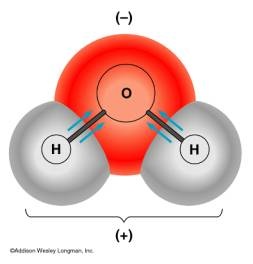
Sometimes the electrons are shared unequally. When they are shared unequally, we use the term polar. Polar - electrons are shared UNEQUALLY.
It is the sharing of electrons!
Sometimes the electrons are shared equally. When they are shared equally, we use the term nonpolar. Nonpolar - electrons are shared EQUALLY.
Sometimes the electrons are shared unequally. When they are shared unequally, we use the term polar. Polar - electrons are shared UNEQUALLY.
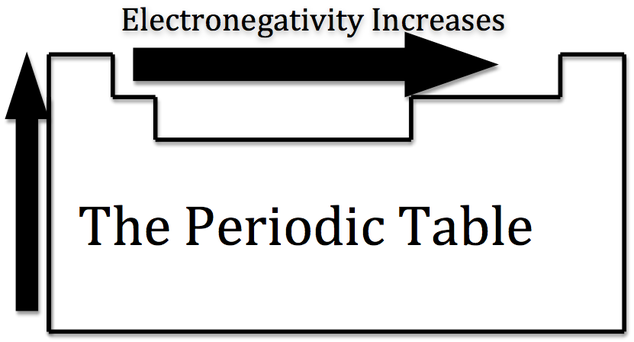
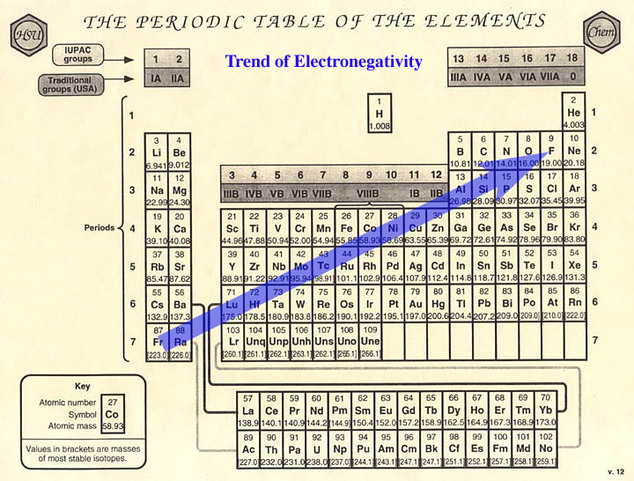
To figure out if it is polar or nonpolar (if you can't tell from just looking at the elements) then you would have to look at the electronegativity.
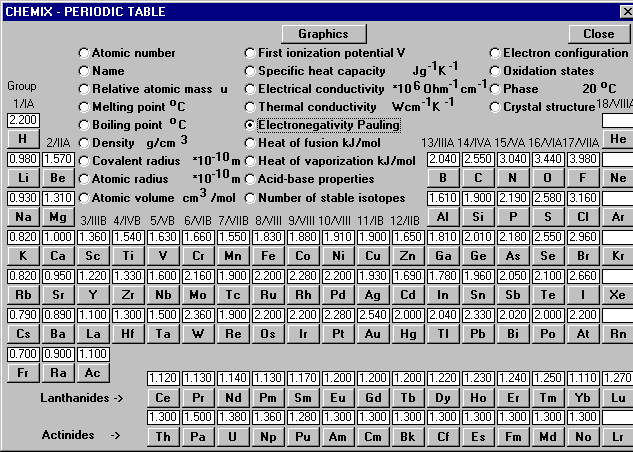
Electronegativity will tell you how attracted an element is to electrons. The higher the electronegativity, the more attracted the element is to electrons. The most electronegative element is fluorine.
Electronegativity will tell you how attracted an element is to electrons. The higher the electronegativity, the more attracted the element is to electrons. The most electronegative element is fluorine.
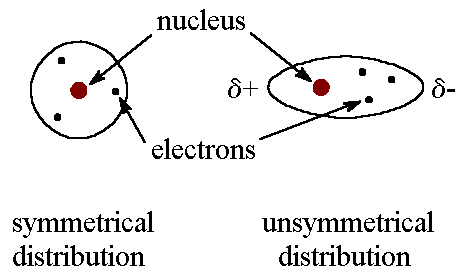
If the elements have electronegativities that are similar to each other, then that means they are sharing similar amount of electrons. If they are sharing about the same amount of electrons we can say that it is nonpolar. If one electronegativity is higher than the other, that means one is sharing more electrons than the other making the sharing of electrons unequal. When it is unequal, it is termed polar. The unequal sharing of electrons will have a partial negative on one end and a partial positive on another.
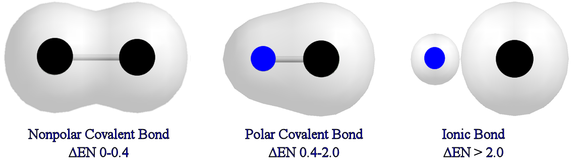
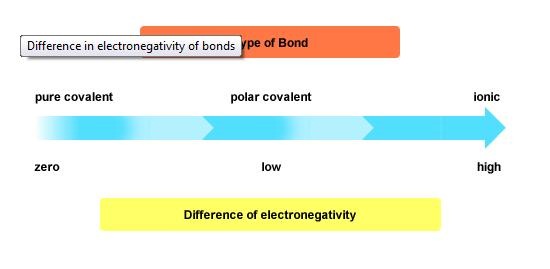
If you can't tell if it's polar or nonpolar, then you would have to look at their electronegativity and compare it. Again, if their electronegativity were similar then it would be nonpolar. If their electronegativity are not, then it would be polar. At times, we can't tell if it's nonpolar or polar. The electronegativity difference chart can tell us for sure if it's nonpolar, polar, or ionic. In order to use this, we need to know their electronegativity and subtract one from another. Mostly we are looking at a difference. The amount difference will tell you what type of bond it is.
Remember to take the absolute value difference! That means if you get a negative value, all you have to do it is change it to a positive value.
Ionic bonds are the strongest. Nonpolar covalent bonds are the weakest.
Ionic bonds are the strongest. Nonpolar covalent bonds are the weakest.
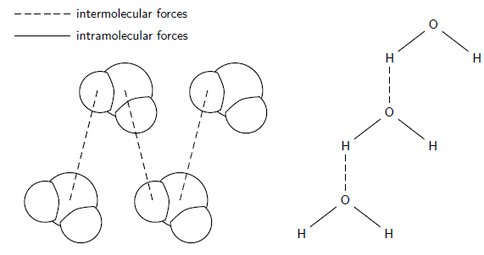
So this whole time we have been learning intramolecular forces. Intramolecular forces are forces within molecules. They are the force that holds together the atoms making up a molecule or compound. Example of an intramolecular force is covalent bonds. Now we are learning about intermolecular forces. Intermolecular force is the attractive force between molecules.
Look at the prefixes.
INTRA - WITHIN (molecules)
INTER - BETWEEN (molecules)
Look at the prefixes.
INTRA - WITHIN (molecules)
INTER - BETWEEN (molecules)
The three intermolecular forces are (starting from strongest to weakest):
-Hydrogen bonding
-Dipole-dipole
-London dispersion
-Hydrogen bonding
-Dipole-dipole
-London dispersion
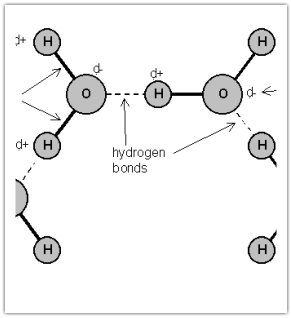
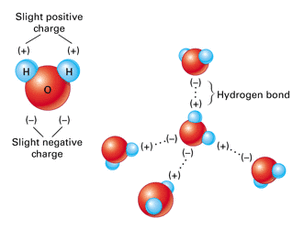
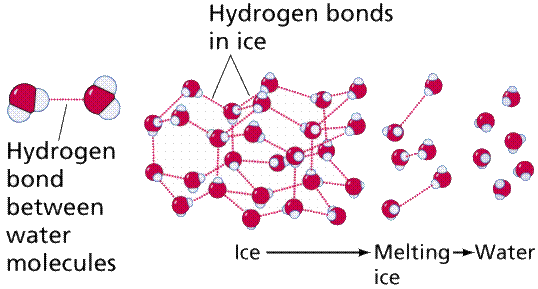
HYDROGEN BONDING
Hydrogen bonding includes H bonded to either N, O, or F. Remember fluorine is the most electronegative element? Well close to that is oxygen and nitrogen. Hydrogen bonding is the strongest force out of the three. Water is known for hydrogen bonding.
Hydrogen bonding includes H bonded to either N, O, or F. Remember fluorine is the most electronegative element? Well close to that is oxygen and nitrogen. Hydrogen bonding is the strongest force out of the three. Water is known for hydrogen bonding.
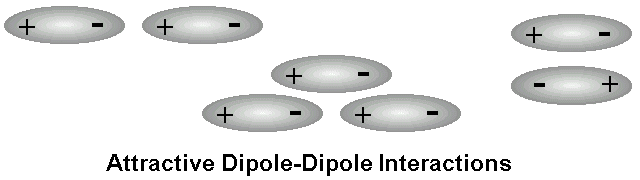
DIPOLE-DIPOLE
Dipole-dipole is the attraction between two dipoles. Every time you see a polar molecule, then it is a dipole-dipole. What is a polar bond again? It is a sharing of unequal electrons. Also, you will have a partial positive and a partial negative.
Dipole-dipole is the attraction between two dipoles. Every time you see a polar molecule, then it is a dipole-dipole. What is a polar bond again? It is a sharing of unequal electrons. Also, you will have a partial positive and a partial negative.
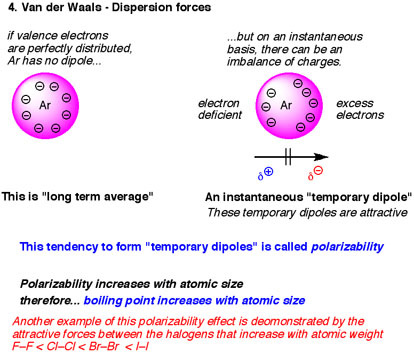
LONDON DISPERSION
London dispersion is the attraction between two instantaneous dipoles. It is the weakest of the three forces. Think of dispersion as constantly moving. Or you can think of London as the London bridge and it's falling down, concluding that London is a weak force. Also, the larger the molecule, the larger the dispersion force. What bond was the weakest when we compared electronegativity? It was nonpolar covalent bonds. So every time you see a nonpolar covalent bond (weak), it has to be a london dispersion force (also weak).
London dispersion is the attraction between two instantaneous dipoles. It is the weakest of the three forces. Think of dispersion as constantly moving. Or you can think of London as the London bridge and it's falling down, concluding that London is a weak force. Also, the larger the molecule, the larger the dispersion force. What bond was the weakest when we compared electronegativity? It was nonpolar covalent bonds. So every time you see a nonpolar covalent bond (weak), it has to be a london dispersion force (also weak).